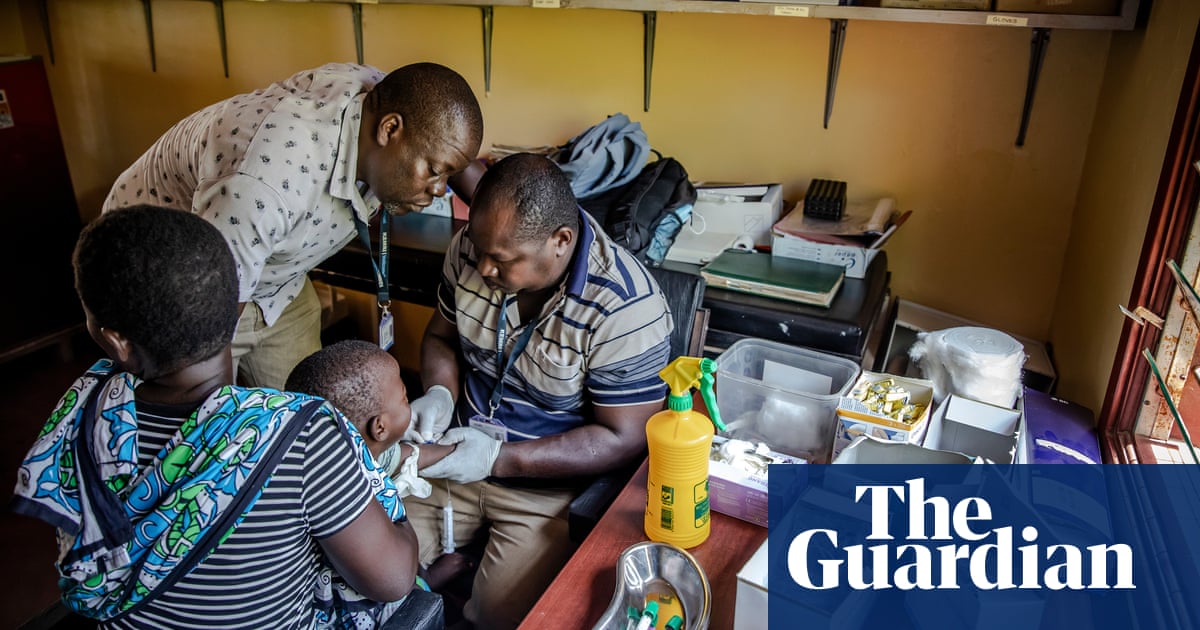
Universal Corporation Limited, a pharmaceutical company based in Kenya, has achieved the distinction of being the first manufacturer in Africa to gain approval from the World Health Organization (WHO) to produce a crucial drug for treating malaria.
Sulfadoxine-pyrimethamine plus amodiaquine (Spaq) is a common medication used to prevent seasonal malaria in children, especially during peak transmission periods like rainy seasons. In the past, African countries have relied on the importation of generic versions of this drug from India and China to meet the demand.
Perviz Dhanani, the managing director of Universal Corporation Limited, stated that the WHO’s pre-qualification is a crucial move in decreasing the dependency on imported medications and enhancing Africa’s ability to independently provide necessary healthcare solutions.
Over 70% of medications utilized in Africa are brought in, and just six out of numerous pharmaceutical companies in Africa have been approved by the WHO.
Some of the barriers to quality local production of drugs include high operation costs, inadequate technical expertise, lack of investment in the pharmaceutical industry, and drug regulation and quality issues. Falsified or substandard antimalarials are estimated to cause up to 116,000 deaths in sub-Saharan Africa every year.
Rephrasing: Receiving approval from the WHO certifying that a company’s manufacturing practices and quality control comply with global standards can facilitate the entry of pharmaceutical companies into new markets and appeal to major purchasers of these treatments, such as donor-funded organizations.
According to the anti-malaria research group Medicines for Malaria Venture (MMV), ensuring that drugs are produced and distributed locally will accelerate the efforts to eradicate malaria. Despite a steady decrease in the number of people living with malaria in Africa over the last 20 years, funding for the disease has remained stagnant in recent years due to donor exhaustion. This has raised concerns among health experts that progress made could be undone.
Dr. André-Marie Tchouatieu, the director of access and product management at MMV, praised drugs such as Spaq for their cost-effectiveness in combating malaria. He stated that their affordability not only improves accessibility, but also promotes self-sufficiency in malaria control, reducing the need for external aid. This allows for strategic allocation of resources to strengthen the overall approach to fighting this destructive disease.
Last year, in Africa’s Sahel region, malaria organizations report that over 48 million children were protected by prevention therapies, such as Spaq, which accounted for nearly 80% of malaria deaths among children under five.
Although malaria vaccines like RTS,S/AS01 are slowly being distributed throughout the continent, access is still restricted and antimalarial medications remain heavily relied upon. Certain research indicates that combining Spaq with vaccines provides greater protection for children under five compared to using only one intervention.
In recent years, there has been a renewed push for Africa to increase its capacity for local production due to distribution issues and vaccine protectionism during the Covid pandemic, which highlighted its weaknesses. Experts in the field of malaria have emphasized the importance of developing a medication that can be readily available in Africa, as it is home to over 95% of global malaria cases and fatalities.
Pierre Hugo, the access director at MMV, stated that the goal is to guarantee a stable supply. Last year, the organization partnered with the Africa Centres for Disease to assist African makers of antimalarial medication in meeting the quality criteria set by the WHO.
MMV and Africa CDC have advocated for the adoption of free trade deals, such as the African Continental Free Trade Area, in African nations. These agreements would facilitate the exchange of manufactured medicines within the region, without imposing tariffs. According to experts, producers of malaria medicine in Africa face tough competition from Indian counterparts who have the advantage of large-scale production. The elimination of trade barriers can greatly impact the success of manufacturing efforts.
Joy Phumaphi, the executive secretary of the African Leaders Malaria Alliance, stated that by receiving support and prioritization from governments, procurement agencies, and other stakeholders, millions of children and pregnant women in Africa could be shielded from malaria.
Source: theguardian.com


